City of Jyväskylä
News
-
Image
 19.4.2024This year, Jyväskylä's May Day will kick off with a bang, when a student-created Vappukuulutus! echoes from the Harju Tower to signal the start of the spring festival week. The song, composed by students and played by Puhkupillit, will be heard from Harju on Wednesday 24 April at 20.00. In addition to the students, Jyväskylä's May Day celebrations offer a wide range of activities for families with children and anyone else interested in the spring festivities.
19.4.2024This year, Jyväskylä's May Day will kick off with a bang, when a student-created Vappukuulutus! echoes from the Harju Tower to signal the start of the spring festival week. The song, composed by students and played by Puhkupillit, will be heard from Harju on Wednesday 24 April at 20.00. In addition to the students, Jyväskylä's May Day celebrations offer a wide range of activities for families with children and anyone else interested in the spring festivities.
-
24.4.2024The city of Jyväskylä wants to hear from residents about their walking experiences and habits in Jyväskylä. What makes you walk and what prevents you from walking? Where do you feel comfortable as a pedestrian and in which places is it difficult to walk? As a resident, you have the best knowledge of how your neighborhood works in terms of walking and how the conditions should be improved. The survey is carried out to support the planning of Jyväskylä's new walking and cycling promotion program. All residents of Jyväskylä can answer the survey, either by themselves or with assistance.Image

-
22.4.2024Kvik kvik, tsirp tsirp and ti-ti-tyy – now you no longer have to guess which bird species it is, as the artificial intelligence of the mobile application recognizes bird species! By making a recording, you can also help science. During the spring, a hundred point counting sites for migratory birds have been set up in the City of Jyväskylä area, where anyone can record birdsong and make their recordings available to researchers. This is the Migratory Bird Spring application developed by the University of Jyväskylä, and this year it will be used to create a point count network together with the City of Helsinki and Metsähallitus National Parks.Image

-
19.4.2024This year, Jyväskylä's May Day will kick off with a bang, when a student-created Vappukuulutus! echoes from the Harju Tower to signal the start of the spring festival week. The song, composed by students and played by Puhkupillit, will be heard from Harju on Wednesday 24 April at 20.00. In addition to the students, Jyväskylä's May Day celebrations offer a wide range of activities for families with children and anyone else interested in the spring festivities.Image

-
12.4.2024The Finnish Sustainable Communities FISU network actively collaborates on projects enhancing resource wisdom. The member municipalities FISU municipalities planned a joint project and shared experiences during the spring meeting of the networkImage

-
30.1.2024Linkki local traffic ticket prices rise in Jyväskylä, Laukaa and Muurame from 1st of February.
Image
-
23.1.2024The City of Jyväskylä, TOYOTA GAZOO Racing World Rally Team, Toyota Mobility Foundation established a new organisation to promote Jyväskylä’s City Planning Projects on January 19, 2024.Image

-
19.1.2024Okko Kamu, who has had a long and distinguished career as a conductor, will be Jyväskylä Sinfonia's next Principal Guest for the next 1.5 years. Kamu began his tenure at the Jyväskylä Sinfonia's opening concert 18.1.2024, where the contract was announced. Kamu also launched the Sibelius Cycle, during which Jyväskylä Sinfonia will perform all of Jean Sibelius' symphonies under rotating conductors over the coming years.Image

-
16.1.2024The guardians of sixth-graders have received a Wilma message with instructions on how to register for seventh grade and the admission practices.Image

-
16.1.2024During week 3, guardians will receive a letter with more detailed instructions on registration practices.Image

-
16.1.2024The city of Jyväskylä's website (www.jyvaskyla.fi) had a user satisfaction survey on 10.10. - 17 December 2023, when a total of 512 respondents participated in the survey.Image

-
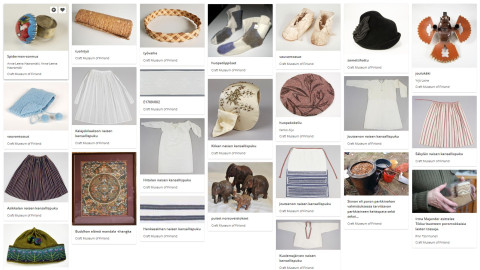
9.1.2024The Craft Museum of Finland is publishing some of its collection materials on the Europeana platform. The platform is a long-standing collaborative project of the member states of the European Union and the European Commission, with the goal of promoting the accessibility and use of European digital cultural heritage for leisure and benefit since its launch in 2008.Image
-
31.12.2023Image






